Dengue NS1 Ag+IgG/IgM Combo Rapid Test
Intended Use
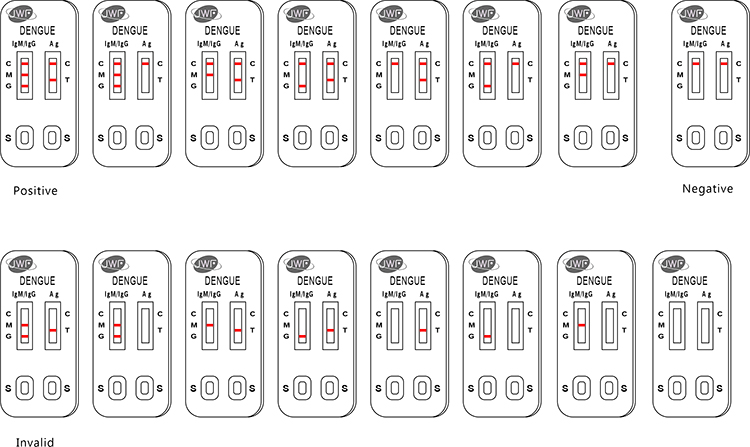
The Dengue Ag(NS1)+IgG/IgM Rapid Test Kit is a lateral flow immunoassay for the simultaneous detection and differentiation of IgG anti–dengue virus, IgM anti-dengue virus and Dengue antigen (Dengue Ag) in human serum, plasma or whole blood samples.
It is intended to be used by the professionals as a screening test and as an aid in the diagnosis of infection with dengue viruses.
For In Vitro Diagnostic use only.