Ebola Rapid Antigen Test
The Ebola Antigen Rapid Test is an in vitro diagnostic
single-use immunoassay for the qualitative detection of antigens from viruses within the Ebolavirus genus but does not differentiate between these viruses.
It is designed for qualitative determination of Ebola antigen in Human whole blood,serum or plasma as an aid in the diagnosis of Ebola infection.
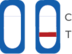
Test Result Interpretation